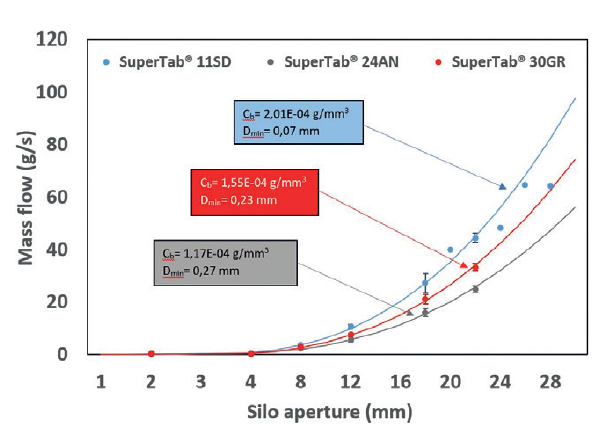
The paracetamol standard powder was sieved using an oscillatory sieve with a 315 μm mesh before use to remove large agglomerates. Copovidone (Kollidon ® VA 64, BASF, Germany) was used as a dry binder and magnesium stearate (Ligastar MG 700, Peter Greven, Germany) as an antiadhesive and lubricant. (Raleigh, NC, USA) and are used as a model drug in the current study. Lactopress ® SD 250, SuperTab ® 11 and SuperTab ® 14 were obtained from DFE Pharma, Germany. Paracetamol standard powder and dense powder were purchased from Mallinckrodt Inc. Dickhoff, Batch versus continuous blending of binary and ternary pharmaceutical powder mixtures, International Journal of Pharmaceutics: X, Volume 4, 2022, 100111, ISSN 2590-1567, ()Īnhydrous lactose ( SuperTab® 21AN), milled lactose monohydrate ( Pharmatose® 200M), spray dried lactose ( SuperTab® 11SD), agglomerated lactose ( SuperTab® 30GR) and microcrystalline cellulose (Pharmacel® 101 and Pharmacel® 102) were obtained from DFE Pharma (Goch, Germany). The result is a product that both flows and compresses well. Janssen, Bernhard Meir, Ralf Weinekötter, Bastiaan H.J. SuperTab® 11SD Spray-dried lactose from DFE Pharma Spray-dried lactose is a finely milled lactose monohydrate which is suspended in water, and spray-dried to give spherical agglomerates of crystalline lactose monohydrate in a matrix of amorphous lactose. Where parameters such as particle size, density and flowability have significant impact on blending performance in a traditional batch process, continuous blending is more robust resulting in uniform blends for a large variety of blend compositions.ĭownload the research paper as PDF: Batch versus continuous blending of binary and ternary pharmaceutical powder mixturesĮffect of coating material on dissolution profile of BCS class 1 anti-epileptic extended-release tabletsĪbout the article: Maarten Jaspers, Sri Sharath Kulkarni, Florian Tegel, Timo P. Therefore, the shape of the particles seems to be a more relevant characteristic to explain the flowability performance of these powders. Sachelac Sorbalac 400 Sorbolac 400 Spherolac Super-Tab SuperTab 11SD SuperTab 21AN Tablettose Tablettose 70 Tablettose 80 Variolac 960 Zeparox EP. Homogeneity of the resulting mixtures is analyzed, which reveals that the impact of material properties is very different in a continuous process. 11SD, SuperTab 30GR, SuperTab 24AN), the SuperTab 11SD has the lowest particle size but also the lowest D min and highest C b. Here, blending of API and excipients is studied in both a batch and a continuous process. Limited information is available on how material properties affect blending performance in a continuous process. The relation between material properties and blend homogeneity, however, is generally based on batch-wise blending trials. With the current shift from batch to continuous manufacturing in the pharmaceutical industry, blending of excipients and API is converted to a continuous process. The material properties of excipients and active pharmaceutical ingredients (API’s) are important parameters that affect blend uniformity of pharmaceutical powder formulations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
